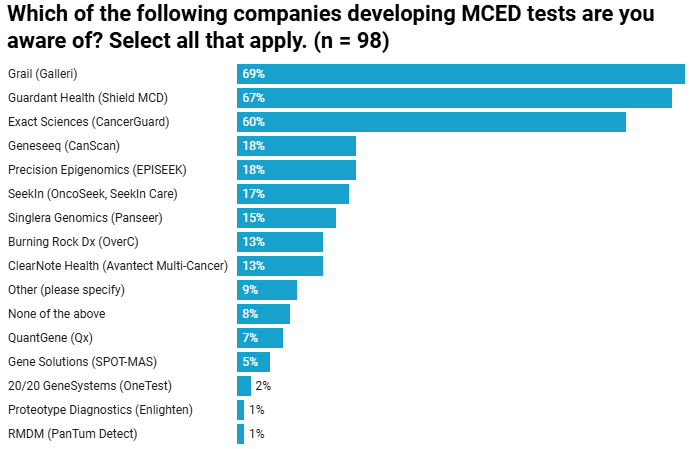
Earlier Cancer Detection.
Reliable Results.
Proactive screening to keep living your best life with earlier multi-cancer detection.*
The EPISEEK™ Test detects the most common cancers before symptoms appear.
EPISEEK™ is a cutting-edge multi-cancer detection (MCD) test that uses a simple blood draw to identify DNA changes linked to over 60 types of cancer. By detecting hypermethylated DNA loci—i.e., regions consistently altered across multiple cancer types—EPISEEK™ provides reliable, early detection, even before symptoms appear.
*The EPISEEK™ test may not identify signals for every type of cancer at every stage. There is a possibility of false positive and false negative results.
A
Adrenal Cortical Carcinoma
Ampulla of Vater
Anus
Appendix, Carcinoma
B
Bile Ducts, Distal
Bile Ducts, Intrahepatic
Bile Ducts, Perihilar
Bladder, Urinary
Bone
Brain
Breast
C
Cervix
Colon and Rectum
E
Esophagus and Esophagogastric Junction
G
Gallbladder
Gastrointestinal Stromal Tumor
Gestational Trophoblastic Neoplasms
K
Kidney
L
Larynx
Leukemia
Liver
Lung
Lymphoma (Hodgkin and Non-Hodgkin)
M
Melanoma of the Skin
Merkel Cell Carcinoma
Mesothelioma, Malignant Pleural
N
Nasal Cavity and Paranasal Sinuses
Nasopharynx
Neuroendocrine Tumors of the Appendix
Neuroendocrine Tumors of the Colon and Rectum
Neuroendocrine Tumors of the Pancreas
O
Oral Cavity
Oropharynx (HPV-Mediated, p16+)
Oropharynx (p16-) and Hypopharynx
Ovary, Fallopian Tube and Primary Peritoneum
P
Pancreas, exocrine
Penis
Plasma Cell Myeloma and Plasma Cell Disorders
Prostate
S
Small Intestine
Soft Tissue Sarcoma of the Abdomen and Thoracic Visceral Organs
Soft Tissue Sarcoma of the Head and Neck
Soft Tissue Sarcoma of the Retroperitoneum
Soft Tissue Sarcoma of the Trunk and Extremities
Soft Tissue Sarcoma Unusual Histologies and Sites
Stomach
T
Testis
U
Ureter, Renal Pelvis
Uterus, Carcinoma and Carcinosarcoma
Uterus, Sarcoma
V
Vagina
Vulva
A
Adrenal Cortical Carcinoma
Ampulla of Vater
Anus
Appendix, Carcinoma
B
Bile Ducts, Distal
Bile Ducts, Intrahepatic
Bile Ducts, Perihilar
Bladder, Urinary
Bone
Brain
Breast
C
Cervix
Colon and Rectum
E
Esophagus and Esophagogastric Junction
G
Gallbladder
Gastrointestinal Stromal Tumor
Gestational Trophoblastic Neoplasms
K
Kidney
L
Larynx
Leukemia
Liver
Lung
Lymphoma (Hodgkin and Non-Hodgkin)
M
Melanoma of the Skin
Merkel Cell Carcinoma
Mesothelioma, Malignant Pleural
N
Nasal Cavity and Paranasal Sinuses
Nasopharynx
Neuroendocrine Tumors of the Appendix
Neuroendocrine Tumors of the Colon and Rectum
Neuroendocrine Tumors of the Pancreas
O
Oral Cavity
Oropharynx (HPV-Mediated, p16+)
Oropharynx (p16-) and Hypopharynx
Ovary, Fallopian Tube and Primary Peritoneum
P
Pancreas, exocrine
Penis
Plasma Cell Myeloma and Plasma Cell Disorders
Prostate
S
Small Intestine
Soft Tissue Sarcoma of the Abdomen and Thoracic Visceral Organs
Soft Tissue Sarcoma of the Head and Neck
Soft Tissue Sarcoma of the Retroperitoneum
Soft Tissue Sarcoma of the Trunk and Extremities
Soft Tissue Sarcoma Unusual Histologies and Sites
Stomach
T
Testis
U
Ureter, Renal Pelvis
Uterus, Carcinoma and Carcinosarcoma
Uterus, Sarcoma
V
Vagina
Vulva
How It Works
EPISEEK™ redefines cancer detection and monitoring, identifying cancer with more specificity than imaging and by being more accessible than invasive tissue biopsies.

Provider Draws Blood

Test Is Sent to Lab

Specimen Is Analyzed

Provider Explains Results
Results in Just 7–10 Days
Who Is EPISEEK™ For?
The EPISEEK™ test is recommended for the following :
Individuals at Higher Risk
- Those with a personal or family history of cancer
- Individuals with lifestyle or environmental exposures that increase risk (e.g., firefighters)
Health-Conscious Individuals
- People who prioritize preventive health care and want to take proactive steps to monitor their health
Employers or Wellness Programs
- Organizations aiming to enhance employee health benefits by providing access to advanced health screenings
Individuals Aged 45 or Older
Healthcare Providers
- Physicians, oncologists, and general practitioners seeking advanced diagnostic tools to offer their patients
- Clinics and hospitals that want to integrate cutting-edge cancer screening into their services
Researchers and Institutions
- Institutions interested in leveraging innovation technologies for further studies in early cancer detection
The EPISEEK™ test is not recommended for the following:

Individuals who are pregnant

Individuals undergoing active cancer treatment or those with an active known malignancy